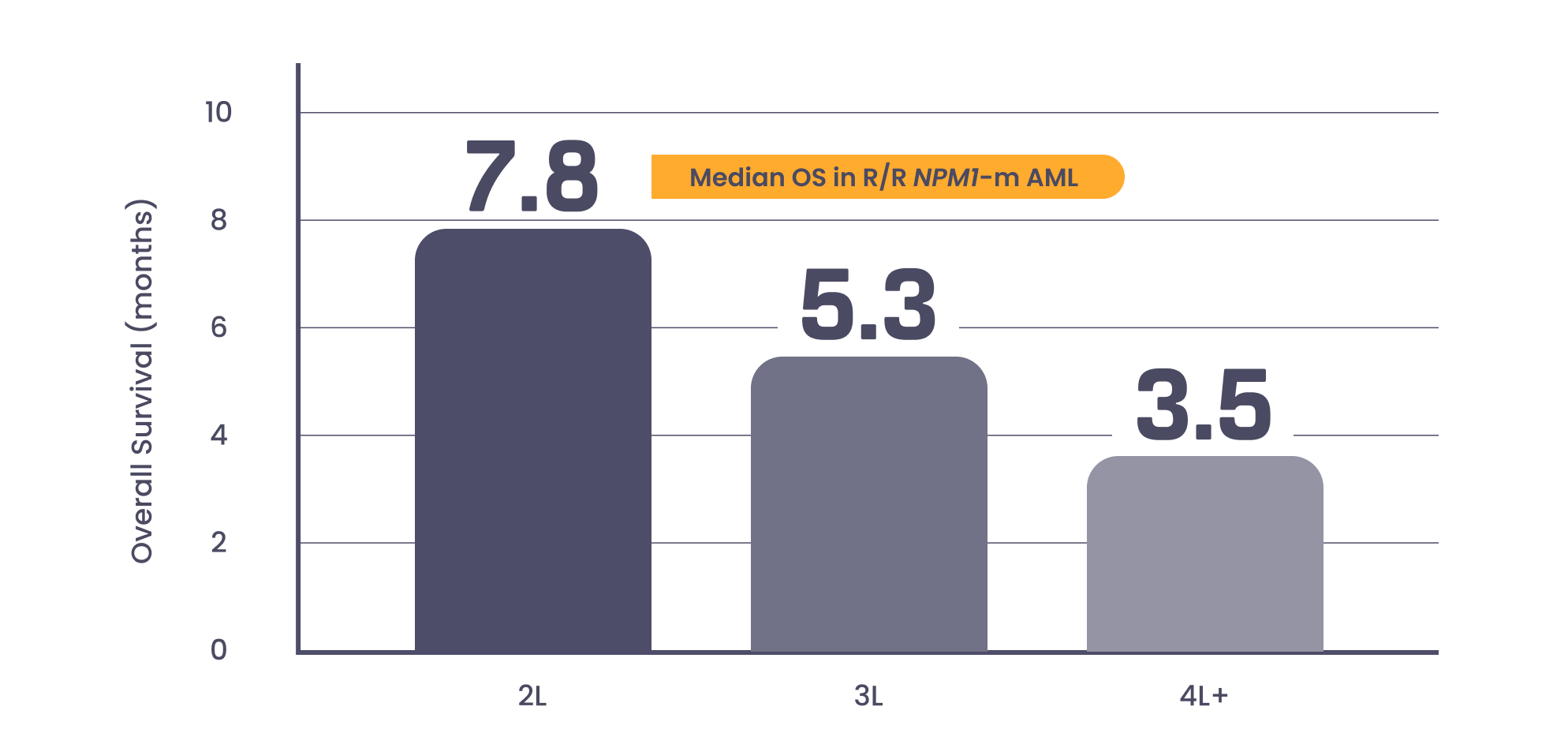
Despite advances, there is considerable unmet need for
patients with R/R NPM1-m AML
Mutated NPM1 is a primary driver of AML1
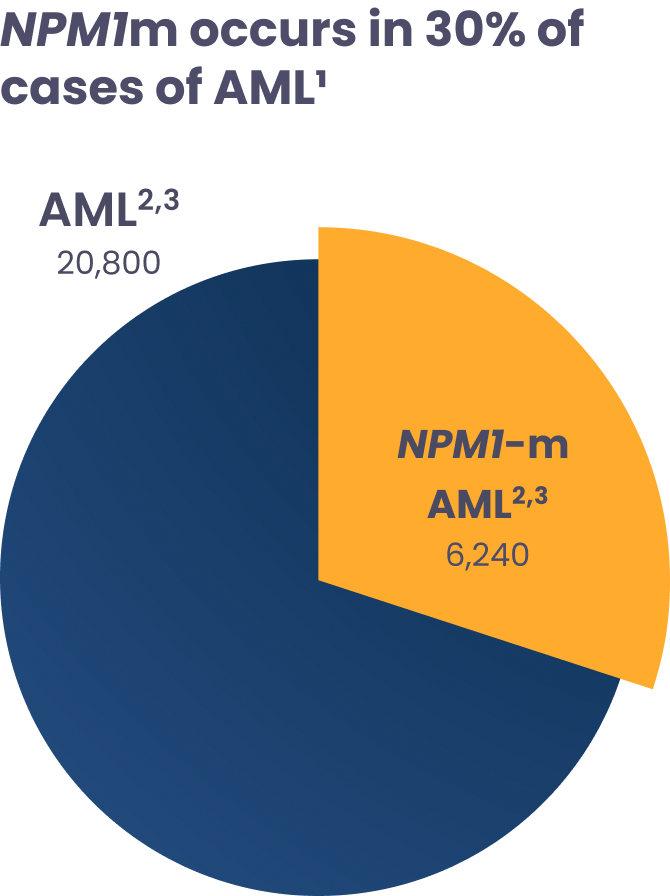
Estimated new cases in the United States in 2024.2,3
NPM1m occurs in 30% of cases of AML1
Common co-mutations
in patients with NPM1m4-6
- FLT3-ITD: 40%
- IDH1/2m: ~15%-30%
- DNMT3Am: 50%
With each relapse, prognosis worsens5
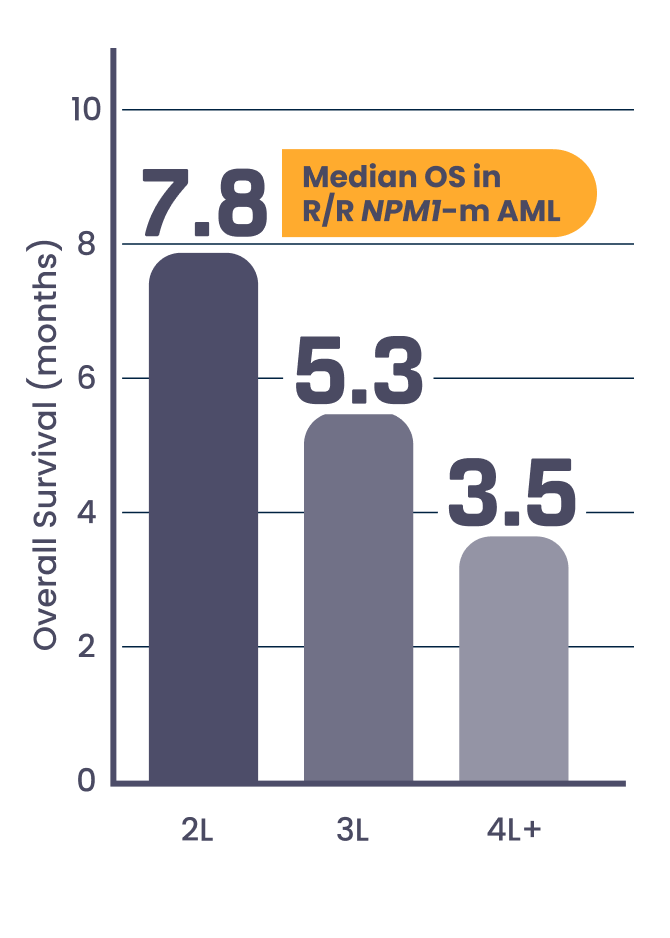
of patients ≥60 years of age with
R/R NPM1-m AML will survive 12 months5,7
aBased on a retrospective analysis of 206 adult patients with relapsed or refractory NPM1-m AML at The University of Texas MD Anderson Cancer Center between September 2012 and December 2020. Various treatment strategies were used. High-intensity regimens included combinations of cytarabine and idarubicin with or without the addition of a nucleoside analog. Low-intensity regimens included either hypomethylating agents or low-dose cytarabine or investigational agents. Low-intensity regimens could include venetoclax from 2018 onward. Targeted therapies were used as single agents or in combination, depending on indication.5
For your appropriate patients
R/R NPM1-m AML is actionable with KOMZIFTI8
AML is a complex, multi-driver disease
- NPM1m is a primary driver of AML1
- NPM1 mutations remain stable at a rate of ~90% throughout the disease course9-11
- NPM1m is often co-mutated with4-6
- FLT3-ITD (40% of patients)
- IDH1/2m (~15%–30% of patients)
- DNMT3Am (50% of patients)
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) recommendation12
Expedited testing for NPM1m at diagnosis and repeat testing at relapse or progression to guide treatment
Available options for mutational status testing include:
- NGS: a full mutational profile of all clinically relevant biomarkers, including NPM112
- PCR: results typically returned within 3-5 days13
See below for a list of labs with a turnaround time of ≤5 days
An FDA-approved test for the detection of NPM1 mutations is not currently available
Consider KOMZIFTI for your appropriate patients with R/R NPM1-m AML8
Download a printable version of this information.
| Lab name | Lab contact information | Lab address | Website |
|---|---|---|---|
| BioReference | 800-229-5227 | 491-B Edward H. Ross Drive, Elmwood Park, NJ 07407 | www.bioreference.com |
| CellNetix | 844-344-4209 | 12501 East Marginal Way S, Suite 200, Tukwila, WA 98168 | www.cellnetix.com |
| Columbia University Department of Pathology and Cell Biology | 212-305-9706 | 630 West 168th Street, P&S 11th Floor, Room 453, New York, NY 10032 | www.pathology.columbia.edu |
| Fred Hutch Cancer Center | 206-667-2592 | Molecular Oncology, Fred Hutchinson Cancer Research Center, 1100 Fairview Avenue N, Rm D2-281, Seattle, WA 98109 | www.fredhutch.org |
| GenPath | 800-627-1479 | 481 Edward H. Ross Drive, Elmwood Park, NJ 07407-0621 | www.genpathdiagnostics.com |
| GoPath Diagnostics | 855-467-2849 | 1000 Corporate Grove Drive, Buffalo Grove, IL 60089 | www.gopathdx.com |
| Hematologics | 206-223-2700 | 3160 Elliott Avenue, Suite 200, Seattle, WA 98121 | www.hematologics.com |
| Invivoscribe | 866-623-8105 | 10222 Barnes Canyon Road, Bldg 1, San Diego, CA 92121 | www.invivoscribe.com |
| UW Medicine | 206-606-7060 | UW Hematopathology Laboratory, G7-800, Fred Hutchinson Cancer Center, 825 Eastlake Avenue E, Seattle, WA 98109 | www.menu.labmed.washington.edu |
| Weill Cornell Medicine | 212-746-2442 | 525 East 68th Street, Room Starr 715, New York, NY 10065 | www.pathology.weill.cornell.edu |
Download a printable version of this information.
Lab name BioReference Lab contact information 800-229-5227 Lab address 491-B Edward H. Ross Drive, Elmwood Park, NJ 07407 Websitewww.bioreference.com |
Lab name CellNetix Lab contact information 844-344-4209 Lab address 12501 East Marginal Way S, Suite 200, Tukwila, WA 98168 Websitewww.cellnetix.com |
Lab name Columbia University Department of Pathology and Cell Biology Lab contact information 212-305-9706 Lab address 630 West 168th Street, P&S 11th Floor, Room 453, New York, NY 10032 Websitewww.pathology.columbia.edu |
Lab name Fred Hutch Cancer Center Lab contact information 206-667-2592 Lab address Molecular Oncology, Fred Hutchinson Cancer Research Center, 1100 Fairview Avenue N, Rm D2-281, Seattle, WA 98109 Websitewww.fredhutch.org |
Lab name GenPath Lab contact information 800-627-1479 Lab address 481 Edward H. Ross Drive, Elmwood Park, NJ 07407-0621 Websitewww.genpathdiagnostics.com |
Lab name GoPath Diagnostics Lab contact information 855-467-2849 Lab address 1000 Corporate Grove Drive, Buffalo Grove, IL 60089 Websitewww.gopathdx.com |
Lab name Hematologics Lab contact information 206-223-2700 Lab address 3160 Elliott Avenue, Suite 200, Seattle, WA 98121 Websitewww.hematologics.com |
Lab name Invivoscribe Lab contact information 866-623-8105 Lab address 10222 Barnes Canyon Road, Bldg 1, San Diego, CA 92121 Websitewww.invivoscribe.com |
Lab name UW Medicine Lab contact information 206-606-7060 Lab address UW Hematopathology Laboratory, G7-800, Fred Hutchinson Cancer Center, 825 Eastlake Avenue E, Seattle, WA 98109 |
Lab name Weill Cornell Medicine Lab contact information 212-746-2442 Lab address 525 East 68th Street, Room Starr 715, New York, NY 10065 |
aThe resource provided was confirmed by reviewing facility websites and/or contacting facilities directly. Kura Oncology, Inc., and Kyowa Kirin Co., Ltd., do not recommend or endorse any facility or lab on this list. It is the sole responsibility of the healthcare professional to select a facility, and this resource is not exhaustive and is only one source of information to assist in that decision. If you would like to suggest a facility be added to the list, please contact Kura Oncology MedInfo at 844-KURAONC.
Summary of 10 labs that include NPM1 as a listed test as of October 2025.
AML, acute myeloid leukemia; DNMT3Am, DNA methyltransferase 3A mutation; FLT3-ITD, FMS-like tyrosine kinase internal tandem duplication; IDH1/2m, isocitrate dehydrogenase 1 and 2 mutation; NCCN, National Comprehensive Cancer Network; NGS, next-generation sequencing; NPM1, nucleophosmin 1; NPM1-m, mutated nucleophosmin 1; NPM1m, nucleophosmin 1 mutation; PCR, polymerase chain reaction; R/R, relapsed or refractory.
References: 1. Falini B et al. Blood Cancer Discov. 2024;5(1):8-20. 2. American Cancer Society. Updated June 5, 2024. Accessed October 22, 2024. https://www.cancer.org/
cancer/types/acute-myeloid-leukemia/about/key-statistics.html 3. Candoni A et al. Hematol Rep. 2024;16(2):244-254. 4. Döhner H et al. Blood. 2017;129(4):424-447. 5. Issa GC et al. Blood Adv. 2023;7(6):933-942. 6. Paschka P et al. J Clin Oncol. 2010;28(22):3636-3643. 7. Issa GC et al. Blood Adv. 2023;7(Suppl):933-942. 8. KOMZIFTI [Prescribing Information]. San Diego, CA; Kura Oncology, Inc. 9. Papadaki C et al. Br J Haematol. 2009;144(4):517-523. 10. Krönke J et al. J Clin Oncol. 2011;29(19):2709-2716. 11. Suzuki T et al. Blood. 2005;106(8):2854-2861. 12. Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Acute Myeloid Leukemia V.2.2026. © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed October 20, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
Discover the efficacy of KOMZIFTI
View the Results From the KOMET-001 Study

WARNING: DIFFERENTIATION SYNDROME
Differentiation syndrome, which can be fatal, has occurred with KOMZIFTI. Signs and symptoms may include fever, joint pain, hypotension, hypoxia, dyspnea, rapid weight gain or peripheral edema, pleural or pericardial effusions, pulmonary infiltrates, acute kidney injury, and rashes. If differentiation syndrome is suspected, interrupt KOMZIFTI, and initiate oral or intravenous corticosteroids with hemodynamic and laboratory monitoring until symptom resolution; resume KOMZIFTI upon symptom improvement.
WARNINGS AND Precautions
Differentiation Syndrome
KOMZIFTI can cause fatal or life-threatening differentiation syndrome (DS). DS is associated with rapid proliferation and differentiation of myeloid cells. Symptoms of DS, including those seen in patients treated with KOMZIFTI, may include fever, hypoxia, joint pain, hypotension, dyspnea, rapid weight gain or peripheral edema, pleural or pericardial effusions, acute kidney injury, and rashes.
In the clinical trial, DS occurred in 29 (26%) of 112 patients with R/R AML with an NPM1 mutation who were treated with KOMZIFTI at the recommended dosage. DS was Grade 3 in 13% and fatal in two patients. In broader evaluation of all patients with any genetic form of AML treated with
KOMZIFTI monotherapy in clinical trials, DS occurred in 25% of patients. Four fatal cases of DS occurred out of 39 patients with KMT2A-rearranged AML treated with KOMZIFTI. KOMZIFTI is not approved for use in patients with KMT2A-rearranged AML.
In the 112 patients with an NPM1 mutation, DS was observed with and
without concomitant hyperleukocytosis, in as early as 3 days and up to
46 days after KOMZIFTI initiation. The median time to onset was 15 days.
Two patients experienced more than one DS event. Treatment was interrupted and resumed in 15 (13%) patients, while it was permanently discontinued in 2 (2%) patients.
Prior to starting treatment with KOMZIFTI, reduce the WBC counts to less than 25 x 109/L. If DS is suspected, interrupt KOMZIFTI, initiate oral or intravenous corticosteroids (e.g., dexamethasone 10 mg every 12 hours) for a minimum of 3 days with hemodynamic and laboratory monitoring. Resume treatment with KOMZIFTI at the same dose level when signs and symptoms improve and are Grade 2 or lower. Taper corticosteroids over a minimum of 3 days after adequate control or resolution of symptoms. Symptoms of DS may recur with premature discontinuation of corticosteroid treatment.
QTc Interval Prolongation
KOMZIFTI can cause QTc interval prolongation. In the clinical trial, QTc interval prolongation was reported as an adverse reaction in 12% of 112 patients treated with KOMZIFTI at the recommended dosage for R/R AML with an NPM1 mutation. QTc interval prolongation was Grade 3 in 8% of patients. The heart-rate corrected QT interval (using Fridericia’s method) (QTcF) was greater than 500 msec in 9% of patients, and the increase from baseline QTcF was greater than 60 msec in 12% of patients. KOMZIFTI dose reduction was required for 1% of patients due to QTc interval prolongation. QTc prolongation occurred in 14% of the 42 patients less than 65 years of age and in 10% of the 70 patients 65 years of age or older.
Correct electrolyte abnormalities, including hypokalemia and hypomagnesemia, prior to treatment with KOMZIFTI. Perform an ECG prior
to initiation of treatment with KOMZIFTI, and do not initiate KOMZIFTI in patients with QTcF > 480 msec. Perform an ECG at least once weekly for the first four weeks on treatment, and at least monthly thereafter. Interrupt KOMZIFTI if the QTc interval is > 500 ms or the change from baseline is > 60 ms (Grade 3). In patients with congenital long QTc syndrome, congestive heart failure, electrolyte abnormalities, or those who are taking
medications known to prolong the QTc interval, more frequent ECG monitoring may be necessary. Concomitant use of KOMZIFTI with drugs known to prolong the QTc interval may increase the risk of QTc interval prolongation, result in a greater increase in the QTc interval and adverse reactions associated with QTc interval prolongation, including Torsades de pointes, other serious arrhythmias, and sudden death.
Embryo-Fetal Toxicity
Based on findings in animals and its mechanism of action, KOMZIFTI can cause embryo-fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment with KOMZIFTI and for 6 months after the last dose. Advise males with female partners of reproductive potential to use effective contraception during treatment with KOMZIFTI and for 3 months after the last dose.
ADVERSE REACTIONS
Fatal adverse reactions occurred in 4 (4%) patients who received KOMZIFTI, including 2 with differentiation syndrome, 1 with infection, and 1 with sudden death. Serious adverse reactions were reported in 79% of patients who received KOMZIFTI. Serious adverse reactions occurring in ≥ 5% of patients included infection without an identified pathogen (29%), febrile
neutropenia (18%), bacterial infection (16%), differentiation syndrome (16%), and dyspnea (6%).
Dosage interruption of KOMZIFTI due to an adverse reaction occurred in
54% of patients. Adverse reactions that required dose interruption in ≥ 2%
of patients included infection without an identified pathogen (15%), differentiation syndrome (13%), febrile neutropenia (5%), pyrexia (4%), electrocardiogram QT prolonged (4%), leukocytosis (4%), bacterial
infection (3%), cardiac failure (2%), cholecystitis (2%), diarrhea (2%),
pruritus (2%), and thrombosis (2%). Dose reduction of KOMZIFTI due to an adverse reaction occurred in 4% of patients. Permanent discontinuation of KOMZIFTI due to an adverse reaction occurred in 21% of patients. Adverse reactions that required permanent discontinuation of KOMZIFTI in ≥ 2% of patients were infection without an identified pathogen (8%), bacterial infection (4%), cardiac arrest (2%), and differentiation syndrome (2%).
Most common (≥ 20%) adverse reactions, including laboratory abnormalities, were aspartate aminotransferase increased (53%), infection without an identified pathogen (52%), potassium decreased (52%), albumin decreased (51%), alanine aminotransferase increased (50%), sodium decreased (49%), creatinine increased (45%), alkaline phosphatase increased (41%), hemorrhage (38%), diarrhea (36%), nausea (35%), fatigue (34%), edema (30%), bacterial infection (28%), musculoskeletal pain (28%), bilirubin increased (27%), potassium increased (26%), differentiation syndrome (26%), pruritus (23%), febrile neutropenia (22%), and transaminases increased (21%).
DRUG INTERACTIONS
Drug interactions may occur when KOMZIFTI is concomitantly used with:
- Strong or Moderate CYP3A4 Inhibitors: Monitor patients more frequently
for KOMZIFTI-associated adverse reactions. - Strong or Moderate CYP3A4 Inducers: Avoid concomitant use of KOMZIFTI.
- Gastric Acid Reducing Agents: Avoid concomitant use of KOMZIFTI with proton pump inhibitors (PPIs), H2 receptor antagonists (H2RAs), or locally acting antacids. If concomitant use with H2RAs or locally acting antacids cannot be avoided, modify KOMZIFTI administration time.
- Take KOMZIFTI 2 hours before or 10 hours after administration of an H2
receptor antagonist. - Take KOMZIFTI 2 hours before or 2 hours after administration of a locally acting antacid.
- Take KOMZIFTI 2 hours before or 10 hours after administration of an H2
- Drugs that Prolong the QT Interval: Avoid concomitant use of KOMZIFTI. If concomitant use cannot be avoided, obtain ECGs when initiating, during concomitant use, and as clinically indicated. Interrupt KOMZIFTI if the QTc interval is > 500 ms or the change from baseline is > 60 ms.
USE IN SPECIFIC POPULATIONS
Pregnancy: Based on findings in animals and its mechanism of action, KOMZIFTI can cause embryo-fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to a fetus. Verify pregnancy status in females of reproductive potential prior to starting KOMZIFTI.
Lactation: Because of the potential for adverse reactions in the breastfed child, advise women not to breastfeed during treatment with KOMZIFTI and for 2 weeks after the last dose.
Infertility: Based on findings in animals, KOMZIFTI may impair fertility in females and males of reproductive potential.
INDICATION
KOMZIFTI is indicated for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (R/R AML) with a susceptible nucleophosmin 1 (NPM1) mutation who have no satisfactory alternative treatment options.
Please see full Prescribing Information, including Boxed WARNING, for additional information.